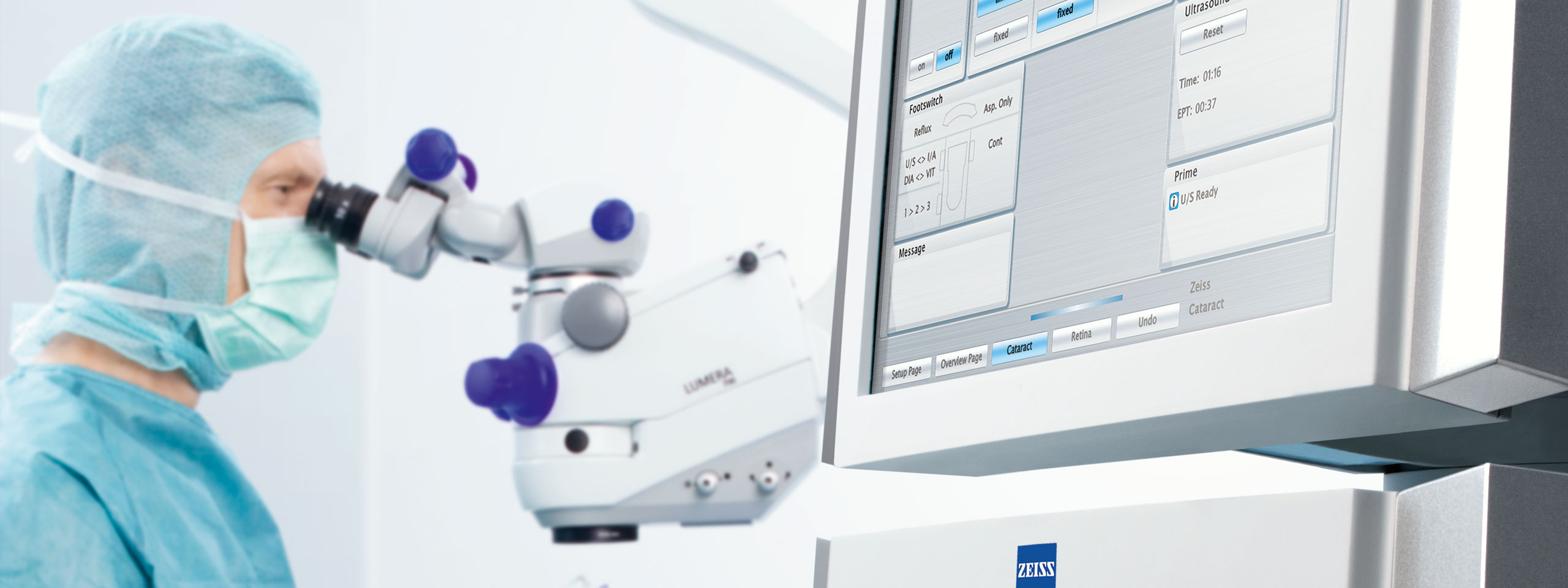
Strategic Medical Device Design & Development
Strategic product design in medical technology, diagnostics and healthcare is part of Milani’s DNA. For over 60 years, we have supported companies in developing medical and diagnostic devices that stand out in the market thanks to their intuitive usability, highest safety standards, and exceptional user experiences.
Whether in medical product design, UX/UI, or circular design, our integrated design approach accelerates your development process and ensures clear differentiation in the market.
Medical Design as a Strategic Tool
In medical and diagnostic technology, design is not something that happens at the end of a project, it starts at the beginning. Whether in hospitals, laboratories, or home care environments, requirements for hygiene, usability, safety, and seamless integration into existing workflows continue to increase. Those who structure and manage this complexity effectively create clear differentiation in the market.
Milani combines deep regulatory expertise with consistently user-centered design and a strong understanding of brand and positioning. We translate your business strategy into a clear, recognizable product language.
With decades of experience, we act as a strategic sparring partner in developing design solutions that bring together safety, usability, and formal clarity, many of them internationally recognized. Our processes are structured and efficient, with defined milestones and tangible outcomes, always focused on time-to-market and design-to-cost.
In addition to industrial design, we develop intuitive UX/UI solutions for connected systems, from embedded interfaces to digital applications.
For Accelerated Medical Device Development
Our User-Centered Design Approach and Usability Engineering
Early assumptions can have a significant impact on the later costs of a project. For this reason, Milani makes concepts testable at an early stage and consistently aligns products with real-world use scenarios, from the initial idea through validation to market implementation.
Interviews, ethnographic research, and hands-on testing form the basis for clear design decisions. We systematically analyze user needs and specific requirements and translate them directly into architecture and interaction concepts. Through iterative prototyping, we validate concepts early and test workflows together with medical professionals, technicians and patients.
You gain reliable decision-making foundations, reduce development risks, and accelerate time-to-market.
Formative and summative testing are integral parts of our process. The results are transparently documented and coordinated closely with R&D, Regulatory, and Product Management. We consistently incorporate MDR, FDA, and IEC 62366 requirements into the design process.
Beyond the product itself, we support companies in developing new offerings within the usage context, including training concepts, repair and service models, and additional patient-focused services.
We connect product, usage, and service in a structured way, developing solutions that convince physicians, nursing staff, and patients alike.

User-Centered Solutions for MDR Compliance
Usability Engineering for Medical Devices
Milani provides user-centered solutions to make your medical devices safe, efficient, and user-friendly. Our experts ensure compliance with MDR and IEC 62366-1 while delivering an excellent user experience through close collaboration between design and development teams.
Regulatory Compliant. Economically Viable.
Ecodesign and Circular Design in Medical Technology
As EU regulations continue to evolve, integrating product sustainability into development in a structured way is becoming increasingly important, particularly in medical technology, where development cycles are long and validation efforts are significant. Regulations such as the Ecodesign for Sustainable Products Regulation (ESPR) and the Packaging and Packaging Waste Regulation (PPWR) are gradually reshaping the framework for manufacturers. At the same time, credible sustainability documentation is increasingly expected throughout the supply chain and in procurement processes.
Even if your company is not yet directly affected, early integration of product sustainability strengthens your market position and provides strategic stability for long-term, resilient product platforms.
Milani supports you in embedding ecodesign and circularity requirements into architectural and specification decisions from the outset. We assess where your product or portfolio is impacted, identify the relevant leverage points, and translate them into solutions that are technically feasible, regulatory compliant, and economically viable.
Furthermore, we view the circular economy as a driver of innovation. Whether through modular product architectures, expanded service offerings, new refurbishment, reuse, or recycling strategies, we help you translate these into viable business models.

End-of-life solutions
Take-back services & circular business models
Take-back, processing, and recycling are becoming increasingly relevant, both from a regulatory and economic perspective.
We help you develop end-of-life solutions and service concepts that fit into your existing structure and create added value. From conceptualizing reverse logistics to designing engaging interaction points for stakeholders.
Extensive experience in medical design and healthcare
Milani has designed medical products and user interfaces across various fields, including:
- Emergency and respiratory care devices
- Diagnostic and monitoring systems
- Ophthalmic and optometric products
- Sterilization equipment
- Cardiothoracic surgery and wound care solutions
- Laboratory and diagnostic instruments
- Hearing aids and audiology devices
- Surgical and medical instruments
- Femtech innovations
- Dental equipment
- Single-use medical products
LET'S TALK
Looking to create innovative, user-centered, and future-proof medical devices? Let’s discuss how Milani can support you.
Example Projects Medical Design
For more than 60 years, Milani has been developing design that reflects the brand's values, optimizes highly specialized processes in medical technology, and inspires doctors, nurses, and patients alike. Be inspired by our projects.